Increase Production Without Increasing Footprint
While your pharmaceutical production and medical device production demands may be increasing, floorspace can come at a premium cost, making expansion of your current process unsustainable.
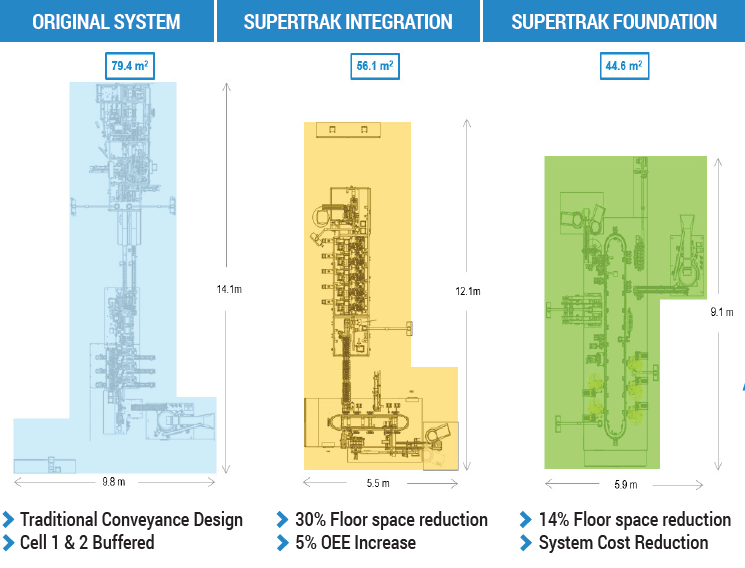
The SuperTrak CONVEYANCE™ platform offers integrated functionality; manufacturers can achieve high-performance in the minimum amount of space by reducing tooling and station redundancies. The platform also offers various configuration options to meet your unique space challenges. One medical device manufacturer was able to gain back 44% of their floorspace by using SuperTrak CONVEYANCE™.
Read more about their story and automation in the life sciences industry here.
Cleanroom Manufacturing
SuperTrak CONVEYANCE™ has been developed over the past 20 years with the medical and pharmaceutical industries in mind.
With few moving parts, the SuperTrak CONVEYANCE™ platform is easy to maintain and generates very little particulate matter, keeping your operations clean and moving quickly. Minimal moving parts lead to low maintenance and results in minimal downtimes and increased throughput.
Quiet Operation
This Smart Conveyance system is also quiet – operating at > 75dBA – making it easier for those working in and around the automation system for long periods of time.
The main reasons that the SuperTrak HORIZON10™ system is quiet are:
- The SuperTrak HORIZON10™ system has very few moving parts. Each shuttle has 4 plastic rollers/wheels that provide low friction and a dampened rolling sound as they ride on the steel rail system.
- The built-in collision avoidance ensures no shuttle-to-shuttle contact or banging noise.
- There are no mechanical stop mechanisms required to precisely stop and locate a shuttle at a process station.
See more frequently asked questions here.



